Sep 13 2017
For several years, Scientists have recognized the vital role played by a molecule called GTP, one of the building blocks of RNA, in the development of cancer and other diseases. However, they had no way to monitor this important nucleotide in live cells, and thus to understand — and interrupt — its contribution to tumor aggressiveness and growth.
Recent research headed by a team from Roswell Park Cancer Institute and published in the Nature Methods journal reports the development of genetically encoded GTP sensors that can quickly identify changes in GTP levels in living cells. The Authors say that it is a finding that could induce the development of innovative approaches for treating an extensive range of solid-tumor cancers, and probably other diseases as well.
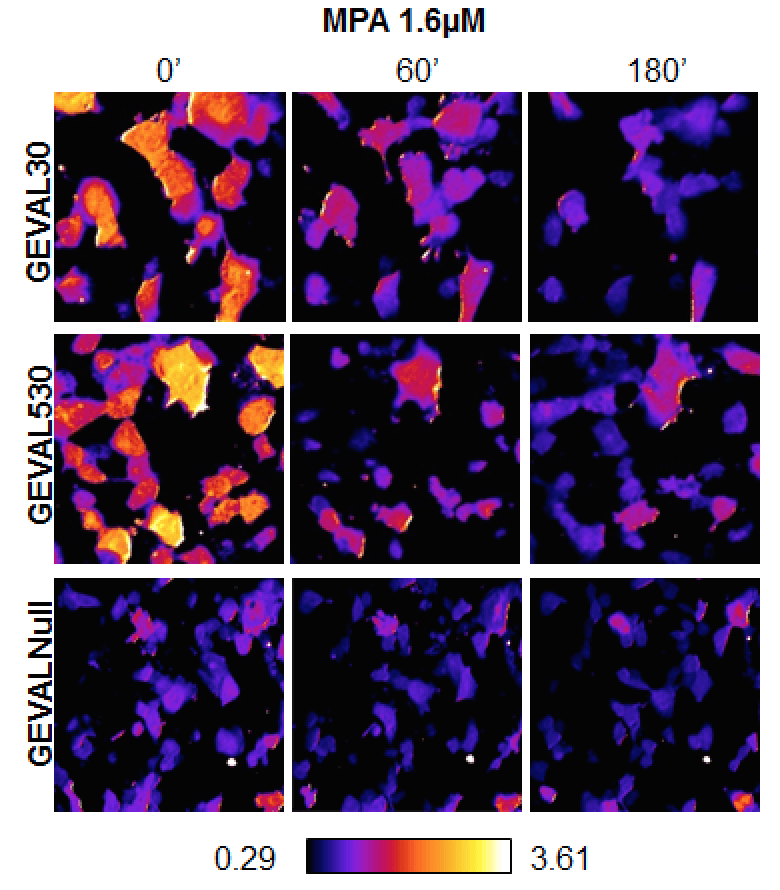 Images from a new fluorescent sensor, showing changes in levels of an important molecule, GTP, following administration of the drug MPA. Credit: Roswell Park Cancer Institute
Images from a new fluorescent sensor, showing changes in levels of an important molecule, GTP, following administration of the drug MPA. Credit: Roswell Park Cancer Institute
The team calls these unique sensors GEVALs, or GTP evaluators. These sensors depend on fluorescent protein technology.
GTP plays an essential role in multiple cellular processes, including protein synthesis, cytoskeleton maintenance and function, nuclear and intracellular transport and intracellular signaling. By inserting a yellow fluorescent protein into a particular bacterial protein and monitoring the comparative change in fluorescence that results when GTP binds to our sensors, we are able to detect and track changes in GTP levels.
Anna Bianchi-Smiraglia, PhD, the Paper’s First Author and a Postdoctoral Researcher, the Department of Cell Stress Biology at Roswell Park
In addition to being the first tool for GTP measurement that can be carried out in live cells, these GEVAL sensors are the first to uncover the spatio-temporal dynamics inducing changes in GTP levels and activity.
Our sensors represent a new and unique tool for assessing changes in GTP levels in cell populations and individual cells, which may in turn point the way to effective strategies for suppressing or even preventing tumor growth. This is an early finding that will have to be further developed through additional research, but it suggests opportunities for developing therapies that interfere with GTP metabolism by targeting key enzymes—perhaps existing therapies as well as new drugs yet to be developed.
Mikhail Nikiforov, PhD, Senior Author of the paper and Professor of Oncology, the Department of Cell Stress Biology at Roswell Park
The collaborators of the team comprise of Scientists from the University of Texas Health Science Center at San Antonio.
This study was supported by grants from the National Cancer Institute (project nos. R21CA151128, R01CA197996, R01CA120244, R01CA193981, R01CA190533, F32CA189622 and F99CA212455), National Institute of General Medical Sciences (project no. R01GM118933), Empire State Development Corp. and Jennifer Linscott Tietgen Family Foundation. The paper, “Internally ratiometric fluorescent sensors for evaluation of intracellular GTP levels and distribution,” can be found at nature.com/nmeth.