Image Credit: Pusan National University
Oxygen plays a key role in various industrial processes, including combustion and energy conversion, which are involved in important fields such as fuel cells, automotive engines, and gas turbines. Thus, a real-time, accurate measurement of oxygen concentration is crucial for the seamless functioning of these industries.
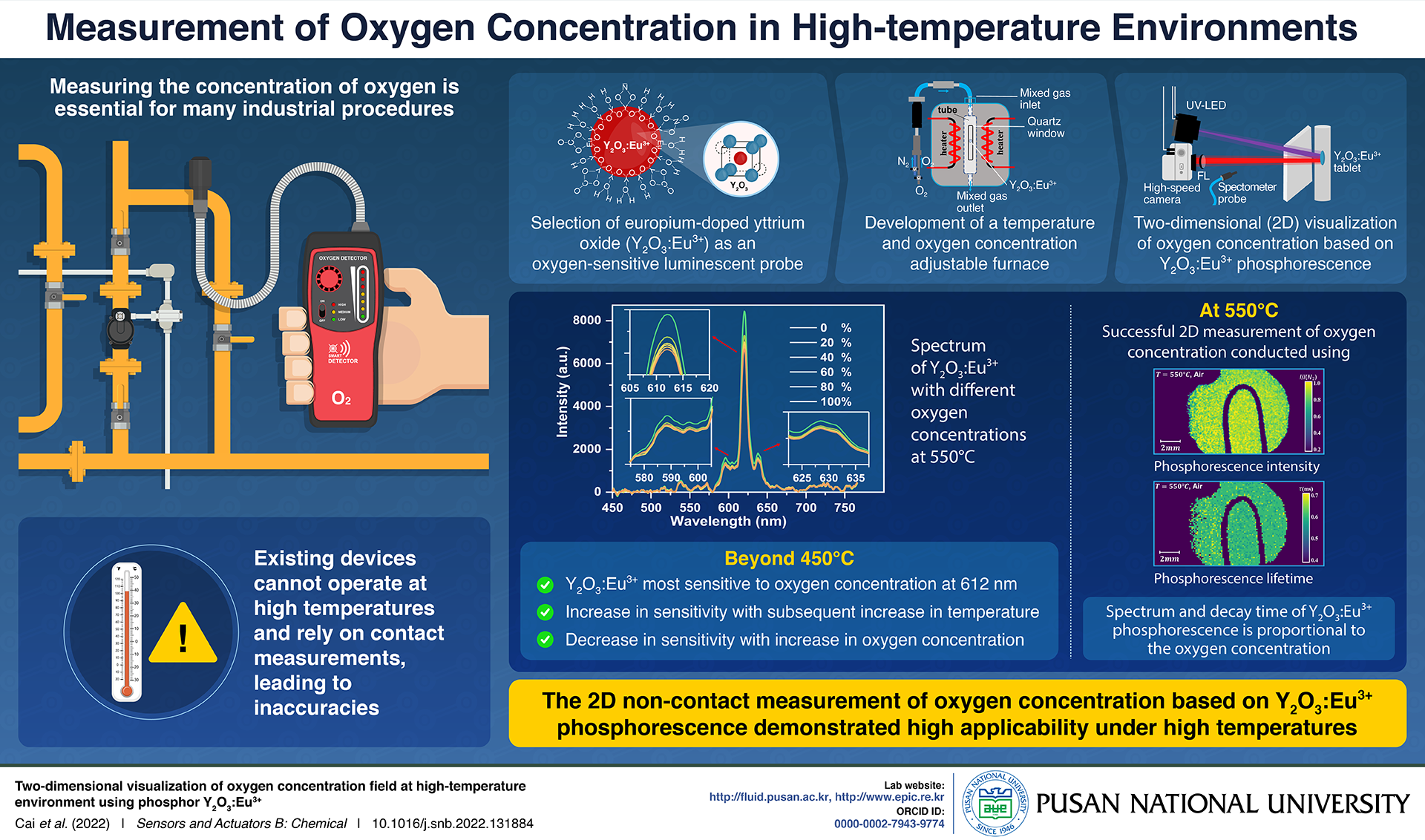
Unfortunately, existing oxygen concentration measurement technologies rely on contact measurements using probes, which cannot withstand high-temperature environments. Moreover, despite the availability of a few optical temperature measurement technologies, the organometallic materials they utilize degrade at temperatures above 120 °C.
To address this problem, a team of researchers led by Prof. Kyung Chun Kim from Pusan National University, Korea, developed and tested a non-contact technique to measure oxygen concentration under high temperatures. In their study, which was made available online on 19 April 2022 and published in Volume 364 of Sensors and Actuators B: Chemical on 01 August 2022, the team described how a phosphorescent material’s glow, or “phosphorescence,” can be leveraged to measure oxygen concentration.
The material in question was yttrium oxide doped with europium (Y2O3:Eu3+)—a phosphor, i.e., a material that emits light in response to radiation—which has a highly temperature-resistant crystalline structure. Like other phosphors, Y2O3:Eu3+ absorbs light energy and re-emits it at a lower frequency. However, owing to its unique molecular arrangement with oxygen vacancies, its phosphorescence varies depending on the surrounding oxygen. This high sensitivity to oxygen makes Y2O3:Eu3+ a suitable non-contact luminescent probe.
To investigate this property further, the team set up a two-dimensional (2D) temperature and oxygen concentration adjustable furnace with a quartz window (a window that allows light to pass freely in both directions) and used it to shine an ultraviolent (UV) LED light towards a Y2O3:Eu3+ tablet. On measuring the resultant phosphorescence using a spectrometer, the team found that it was most sensitive to the oxygen concentration at a temperature beyond 450 °C for a wavelength of 612 nm. Beyond 450 °C, the sensitivity of Y2O3:Eu3+ to oxygen concentration increased with increasing temperature but decreased with an increase in the oxygen concentration.
Importantly, they also observed two properties of Y2O3:Eu3+ phosphorescence that could be used to measure oxygen concentration at 550 °C: its intensity and lifetime, i.e., the time it takes for Y2O3:Eu3+ to stop emitting light. Although measurements using the latter were slightly more accurate, these findings demonstrated the overall applicability of using the phosphorescence of Y2O3:Eu3 at high temperatures.
Discussing these findings, Dr. Kim states, “Our study is the first to develop a simple, non-contact, 2D method that can provide technical support for the performance improvement of many industrial products at high temperatures.”
What are the implications of these findings? Prof. Kim further remarks, “This method can enhance basic mechanism research and industrial production applications, which would help us understand unknown thermophysical phenomena in daily life and engineering.”
It is safe to say that by virtue of its simplicity and accuracy, this method is likely to find a home in many fields and pave the way for a new era of oxygen concentration sensors!