Jul 4 2016
A team of researchers at Tokyo Medical and Dental University (TMDU) demonstrated that an analyzer recently created to quantify blood coagulability has the sensitivity to identify hypercoagulatibility connected with stroke risk in individuals without atrial fibrillation.
 Assessing embolic stroke risk by measuring blood coagulability. (Credit: Department of Biofunctional Informatics, TMDU)
Assessing embolic stroke risk by measuring blood coagulability. (Credit: Department of Biofunctional Informatics, TMDU)
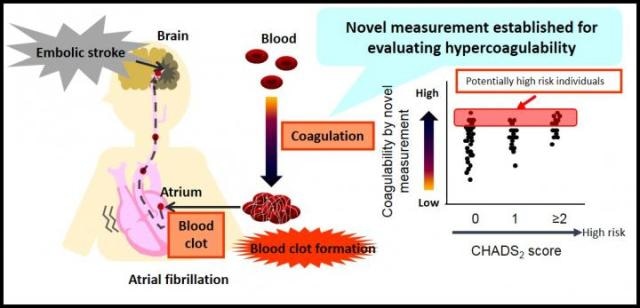
Atrial fibrillation (AF) causes an unusual and sometimes rapid heart rate, and is a typical risk factor for stroke. The CHADS2 predictive score is utilized to determine this risk in AF patients and establish the necessity for anticoagulation therapy. Since a few sections of this score are also connected with atherosclerosis risk and increased blood coagulability, a high score has been suggested as connected to hypercoagulability, which refers to an increased tendency for blood clotting, in both non-AF and AF patients. This association however has not been completely examined, partially because of the absence of a sensitive method for identification. A research group from TMDU utilized an extremely sensitive technique to quantify minute changes in blood coagulation, and discovered hypercoagulability in non-AF patients with high CHADS2 scores. The study was lately reported in PLOS ONE.
A number of chemical and physical factors influence blood clotting, some of which can be determined over time to decide blood coagulability and the probability of clot formation. A recently developed test known as dielectric lood coagulometry (DBCM) helps measuring changes in the dielectric permittivity of whole blood, characterizing clumping of red blood cells. The researchers utilized DBCM to identify changes in the dielectric permittivity of whole blood at 10 MHz. Correlations between untreated blood and that with additional heparin (very thin blood) or tissue factor (a blood-clotting accelerator) helped obtaining of a coagulability index.
We calculated the end of acceleration time (EAT) as an index of coagulability from temporal changes in dielectric permittivity. This value reduced when tissue factor was added, and increased with heparin present. It was also sensitive enough to detect small changes in coagulability, particularly in hypercoagulability.
Coauthor Satomi Hamad
EAT also boasts high reliability and reproducibility.
The researchers discovered that patients accepting warfarin had a considerably longer EAT than those without, thus assuring the anticoagulation impact. They additionally demonstrated that patients with a high CHADS2 score had a considerably shorter EAT that characterized hypercoagulability compared with patients possessing lower CHADS2 scores.
Intriguingly, EAT varied widely in patients with CHADS2 scores of 0 or 1. This suggests that DBCM can identify high risk of thrombosis even in patients with low CHADS2 scores.
Lead author Yuki Hasegawa